3D multicolor super-resolution microscopy of thick samples
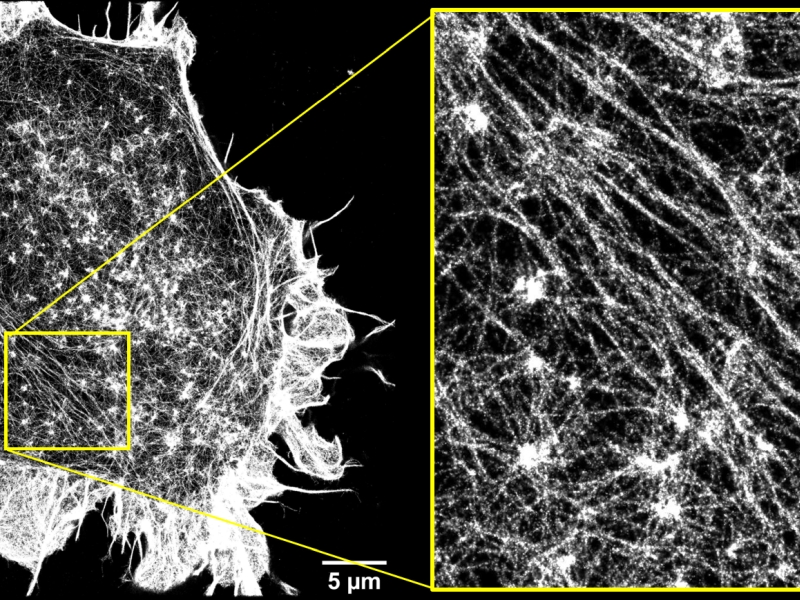
Superresolution 2D STORM image of the actin cortex of HEK cells.
Description
Super-resolution fluorescence microscopy allows to resolve details one order of magnitude smaller than the optical resolution limit, without having to resort to methods such as electron or scanning probe microscopy techniques. Among the many available super-resolution techniques, the highest resolution has been achieved by localization-based methods such as STochastic Optical Reconstruction Microscopy (STORM) and PhotoActivated Localization Microscopy (PALM), which rely on chemical control over fluorophore photophysics in order to resolve the spatial position of individual fluorescent molecules with nanometer accuracy. In our lab we set up novel optical tecniques to achieve improved three-dimensional super-resolution images through a combination of inclined illumination and astigmatism to acquire volumetric super-resolved images of thick samples. We apply 2D and 3D STORM microscopy to study the structure of the actin cortex of eukaryotic cells and how it is regulated by the extracellullar mechanical environment. Moreover, PALM microscopy is applied to the sytudy of membrane proteins distribution on planktonic bacteria and bacteria biofilms to investigate the molecular mechanisms at the basis of antibiotic resistance. Lastly, we adopt a combination of single.molecule three-dimensional tracking and super-resolution imaging to study the dynamics of receptors and transcription factors in living cells.
INO Staff
Gardini Lucia (Contact Person)
Personnel not belonging to INO
Prof. Francesco S. Pavone
Prof. Marco Capitanio
Chiara Caldini, PhD
Giulia Senesi (PhD student)

